A-B-C Blog
Top 10 Facts About 2 Bromo 1 Phenyl Pentan 1 One You Should Know?
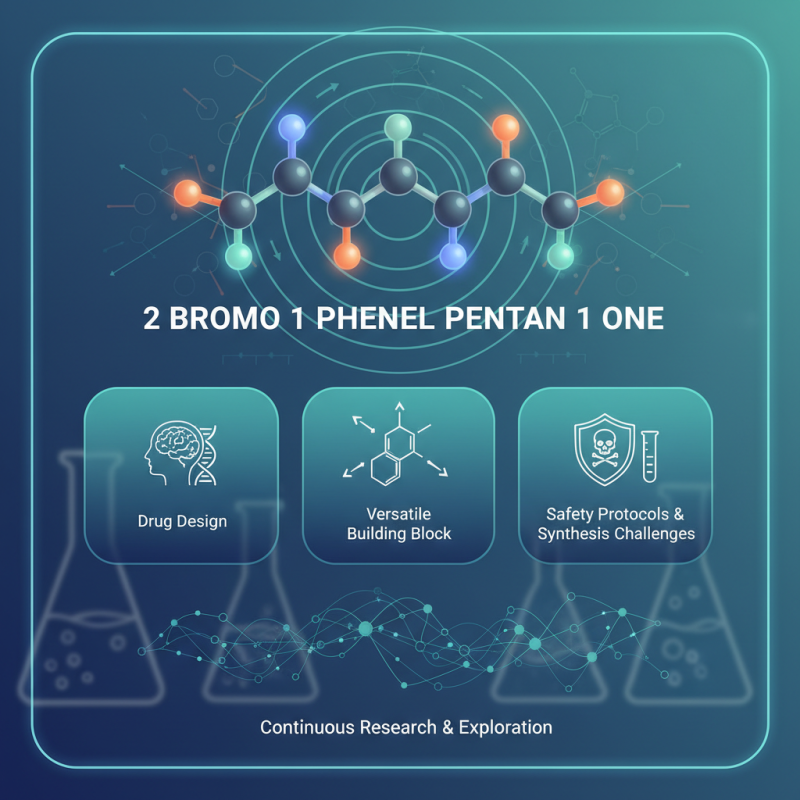
In the realm of organic chemistry, 2 bromo 1 phenyl pentan 1 one stands out for its unique properties. Dr. Sarah Thompson, a leading expert in synthetic organic compounds, emphasizes its significance by stating, "2 bromo 1 phenyl pentan 1 one presents intriguing possibilities in drug design." This compound holds potential that researchers are only beginning to explore.
Recent studies reveal that this compound can serve as a versatile building block in various chemical reactions. Its structure allows for multiple functionalizations, making it valuable for creating diverse derivatives. However, the practical applications and safety protocols surrounding 2 bromo 1 phenyl pentan 1 one require careful consideration. Despite its potential, navigating the complexities of its synthesis and application can be challenging.
The journey of understanding 2 bromo 1 phenyl pentan 1 one invites discussion. As scientists delve deeper, they often face unexpected hurdles, underscoring the need for continuous research. A thorough exploration of its properties and implications is essential for harnessing its full potential while ensuring safety in all applications.
Chemical Structure and Composition of 2 Bromo 1 Phenyl Pentan 1 One
2 Bromo 1 Phenyl Pentan 1 One is an interesting compound. It has a distinct chemical structure that includes both bromo and phenyl groups. The presence of the bromo atom enhances its reactivity. This compound's structure contributes to its various applications in research.
The molecular formula can be denoted as C11H13BrO. This indicates the presence of carbon, hydrogen, bromine, and oxygen. The arrangement is vital for understanding its chemical behavior. It is characterized by a pentan-1-one chain, which is a five-carbon straight chain with a ketone group. This structure affects its physical properties and interactions.
While its features may seem simple, they lead to complex behaviors. The compound can be unstable under certain conditions. This highlights the importance of careful handling. Researchers must consider these aspects when using the substance. Each factor plays a role in its practical applications, making it a subject worthy of further study.
Synthesis Methods and Procedures for 2 Bromo 1 Phenyl Pentan 1 One
Synthesis of 2 Bromo 1 Phenyl Pentan 1 One involves several steps. Starting with a suitable phenyl-containing substrate is crucial. The choice of starting materials impacts the overall yield and purity. Reaction conditions also play a significant role. Temperature and solvent selection can alter outcomes dramatically. For this compound, controlling the reaction environment is vital.
One common method begins with bromination. This step introduces bromine into the molecular structure. Typically, this is done using bromine and a suitable catalyst. The next stage involves reduction, converting the intermediate product into the desired compound. This process may require trial and error. Yield may vary, and sometimes, unexpected byproducts emerge.
Troubleshooting synthesis often leads to difficulties. Variations in reaction time can affect concentration. Monitoring the reaction closely is necessary but often overlooked. Documenting each phase helps in determining optimal parameters for future attempts. Lab notes should include all observations, including failures. This way, improvements can be made in subsequent synthesis runs.
Applications and Uses of 2 Bromo 1 Phenyl Pentan 1 One in Industry
2 Bromo 1 Phenyl Pentan 1 One is a compound with exciting industrial applications. This chemical is often utilized in the production of various pharmaceuticals. Its unique structure allows it to act as an essential intermediate. Manufacturers appreciate its effectiveness in synthesizing other complex compounds.
In addition, 2 Bromo 1 Phenyl Pentan 1 One serves an important role in chemical research. Scientists use it to test reactions and develop new methodologies. Its properties can lead to the discovery of innovative solutions. However, the challenges in handling this compound are noteworthy. Proper precautions are crucial during its usage.
Some industries have found it useful in agrochemicals as well. It enhances crop protection products. Yet, the long-term environmental impact remains a topic of debate. Balancing efficiency with safety is vital. As more researchers explore this compound, we must reflect on its broader implications. Careful consideration is essential in every step of its application.
Safety and Handling Precautions for 2 Bromo 1 Phenyl Pentan 1 One
When working with 2 Bromo 1 Phenyl Pentan 1 One, safety should be your top priority. This chemical compound can pose several health risks if not handled correctly. According to the Centers for Disease Control and Prevention (CDC), proper personal protective equipment (PPE) is essential. Think gloves, goggles, and lab coats. Always ensure adequate ventilation in your workspace to minimize inhalation risks.
One report from the Occupational Safety and Health Administration (OSHA) notes that exposure can lead to respiratory issues and skin irritation. Inhaling vapors can cause dizziness. Always keep this chemical in a tightly sealed container. Store it in a cool, dry place away from direct sunlight. Label containers clearly to avoid confusion.
Tips: Always conduct a risk assessment before starting your work. Familiarize yourself with Material Safety Data Sheets (MSDS) for detailed handling instructions. In case of a spill, have an emergency response plan ready. Keep a first aid kit nearby for quick access. Regularly inspect your PPE for wear and tear. Protect yourself and your environment diligently.
Regulatory Status and Environmental Impact of 2 Bromo 1 Phenyl Pentan 1 One
The regulatory status of 2 Bromo 1 Phenyl Pentan 1 One varies globally. In many regions, it is classified as a controlled substance due to its chemical properties and potential applications. According to a 2021 report by the European Chemicals Agency, compounds with bromine are often subject to stringent regulations. This classification helps to mitigate risks related to misuse and environmental impact.
The environmental impact of this compound is significant. Research indicates that brominated compounds can persist in the environment, affecting soil and water quality. A report from the U.S. Environmental Protection Agency highlights concerns over their toxicity to aquatic life. Consequently, understanding and managing these chemicals is crucial for protecting ecosystems and public health.
Tips: Always stay informed about the regulatory updates concerning chemical substances. Regularly check the latest reports from environmental agencies. Awareness can lead to better practices in handling and disposing of such materials safely. Balancing the benefits of chemical applications with potential risks is essential. Reflecting on our practices can lead to better outcomes for society and the environment.